Evaporation is an important concept taught in the topic of Heat Energy.
Today, I’d like to discuss some answering techniques that can be used to tackle questions on Evaporation.
What Your Child Needs To Know
At the primary school level, evaporation is known as the process by which water changes from a liquid state (water) to a gaseous state (water vapor).
Read Also
The main difference between evaporation and boiling is that evaporation occurs at all temperatures but boiling only occurs at 100˚C.
There are four factors of evaporation, namely:
- W – Presence of Wind
- H – Humidity
- A – Exposed Surface Area
- T – Temperature
Question
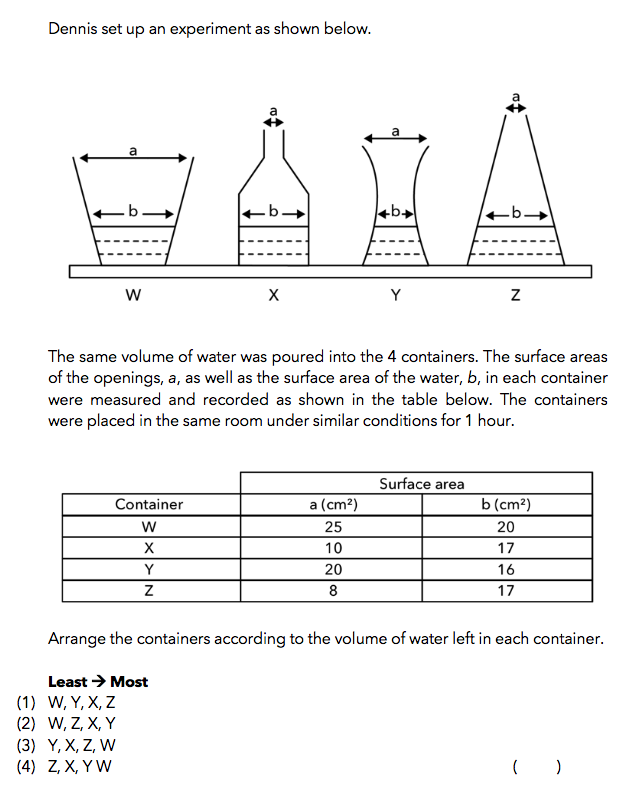
Thought Process
In the above question, we’re looking specifically at the effect of exposed surface area on the rate of evaporation.
This question requires students to arrange the containers according to the volume of water left in each container, from the least to the most.
This means that students have to arrange them based on the order of descending rates of evaporation.
One important point to note is that the rate of evaporation is dependent on the exposed surface area of the water in contact with the air, and NOT the size of the mouth of the containers.
Thus, the focus should only be on the value of “b”, which is the surface area of the water exposed to air.
With the above thought process in mind, we are able to observe that the container with the largest “b” value is W and the container with the smallest “b” value is Y. This means that the answer would be W, __ , __ , Y.
Next Step
In order to fill in the two blanks above with X and Z, we will need to shift our focus to the shape of the container.
The exposed surface area of the water in container X will not change as the water level decreases because of its straight walls. Thus, the rate of evaporation of the water in Container X is constant.
In contrast, the walls of Container Z is slanted. This means that the exposed surface area of the water increases as the water level decreases. This suggests that the rate of evaporation in Container Z would increase over time.
Thus, the rate of evaporation of water in container Z is faster than the rate of evaporation of water in container X.
Final Answer
Thus, the answer to this question is Option (2) – W, Z, X, Y.

If you like our methodology, we’ve some upcoming workshops:







